This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, which are subject to the “safe harbor” created by those sections for such statements. For more information about Omeros and its programs, visit Forward-Looking Statements One of these novel targets, GPR174, modulates a new cancer immunity axis recently discovered by Omeros, and the company is advancing GPR174-targeting antibodies and small-molecule inhibitors. Omeros’ pipeline holds a diverse group of preclinical programs including a proprietary-asset-enabled antibody-generating technology and a proprietary GPCR platform through which it controls 54 GPCR drug targets and their corresponding compounds. OMS906, Omeros’ inhibitor of MASP-3, the key activator of the alternative pathway of complement, is in a Phase 1 clinical trial, and the company’s PDE7 inhibitor program OMS527, targeting addiction and movement disorders, has successfully completed a Phase 1 trial.   
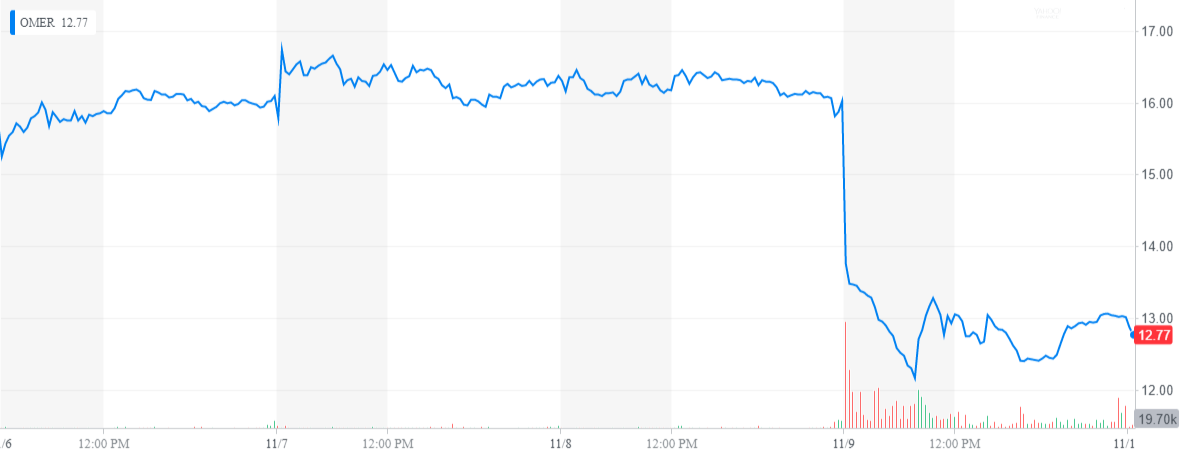
Narsoplimab is also in multiple late-stage clinical development programs focused on other complement-mediated disorders, including IgA nephropathy, atypical hemolytic uremic syndrome and COVID-19. Omeros’ lead MASP-2 inhibitor narsoplimab targets the lectin pathway of complement and is the subject of a biologics license application under priority review by FDA for the treatment of hematopoietic stem cell transplant-associated thrombotic microangiopathy. Its commercial product OMIDRIA ® (phenylephrine and ketorolac intraocular solution) 1%/0.3% continues to gain market share in cataract surgery. Omeros is a commercial-stage biopharmaceutical company committed to discovering, developing and commercializing small-molecule and protein therapeutics for large-market and orphan indications targeting inflammation, immunologic diseases (e.g., complement-mediated diseases and cancers) and central nervous system disorders. The BLA for narsoplimab in HSCT-TMA was accepted for filing in January 2021 under FDA’s Priority Review program. It has Breakthrough Therapy and Orphan designations in both HSCT-TMA and IgA nephropathy. Narsoplimab is the first drug candidate submitted to FDA for approval in HSCT-TMA. Omeros is evaluating potential next steps as it awaits additional information from FDA and plans to obtain FDA approval for narsoplimab in HSCT-TMA, a frequently lethal complication of HSCT for which there is no FDA-approved treatment, as quickly as possible. FDA stated that the notification does not reflect a final decision on the information under review.įDA did not provide specific details of the deficiencies in its notification however, in a meeting held on September 30, 2021, FDA expressed its intention to work with Omeros to resolve any issues as expeditiously as possible, but the company does not currently expect any such resolution to occur by the Octotarget action date under the Prescription Drug User Fee Act (PDUFA). Food and Drug Administration (FDA) notified the company that, as part of FDA’s ongoing review of the company’s Biologics License Application (BLA) for narsoplimab in the treatment of hematopoietic stem cell transplant-associated thrombotic microangiopathy (HSCT-TMA), FDA has identified deficiencies that preclude discussion of labeling and post-marketing requirements/commitments at this time. Omeros Corporation (Nasdaq: OMER) today announced that the U.S.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
